Flame temperatures of magnesium and magnesium alloys can reach 3,100 ☌ (5,610 ☏), although flame height above the burning metal is usually less than 300 mm (12 in). Magnesium is highly flammable, especially when powdered or shaved into thin strips, though it is difficult to ignite in mass or bulk. Magnesium also reacts exothermically with most acids such as hydrochloric acid (HCl), producing the metal chloride and hydrogen gas, similar to the HCl reaction with aluminium, zinc, and many other metals. Magnesium's reversible reaction with water can be harnessed to store energy and run a magnesium-based engine. The reaction occurs faster with higher temperatures (see § Safety precautions). When submerged in water, hydrogen bubbles form slowly on the surface of the metal, and if powdered, it reacts much more rapidly. 
Magnesium reacts with water at room temperature, though it reacts much more slowly than calcium, a similar group 2 metal. However, this oxide may be combined with hydrogen peroxide to form magnesium peroxide, MgO 2, and at low temperature the peroxide may be further reacted with ozone to form magnesium superoxide Mg(O 2) 2. 
It tarnishes slightly when exposed to air, although, unlike the heavier alkaline earth metals, an oxygen-free environment is unnecessary for storage because magnesium is protected by a thin layer of oxide that is fairly impermeable and difficult to remove.ĭirect reaction of magnesium with air or oxygen at ambient pressure forms only the "normal" oxide MgO. However, this reaction is much less dramatic than the reactions of the alkali metals with water, because the magnesium hydroxide builds up on the surface of the magnesium metal and inhibits further reaction. When finely powdered, magnesium reacts with water to produce hydrogen gas: The malleability of polycrystalline magnesium can also be significantly improved by reducing its grain size to ca. It becomes much more malleable when alloyed with small amounts of other metals, such as 1% aluminium. 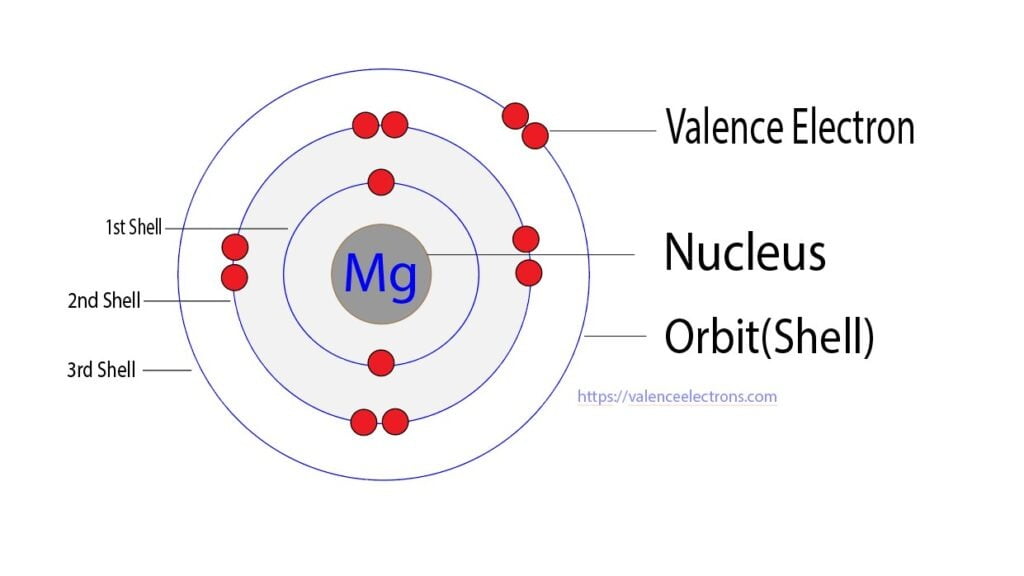
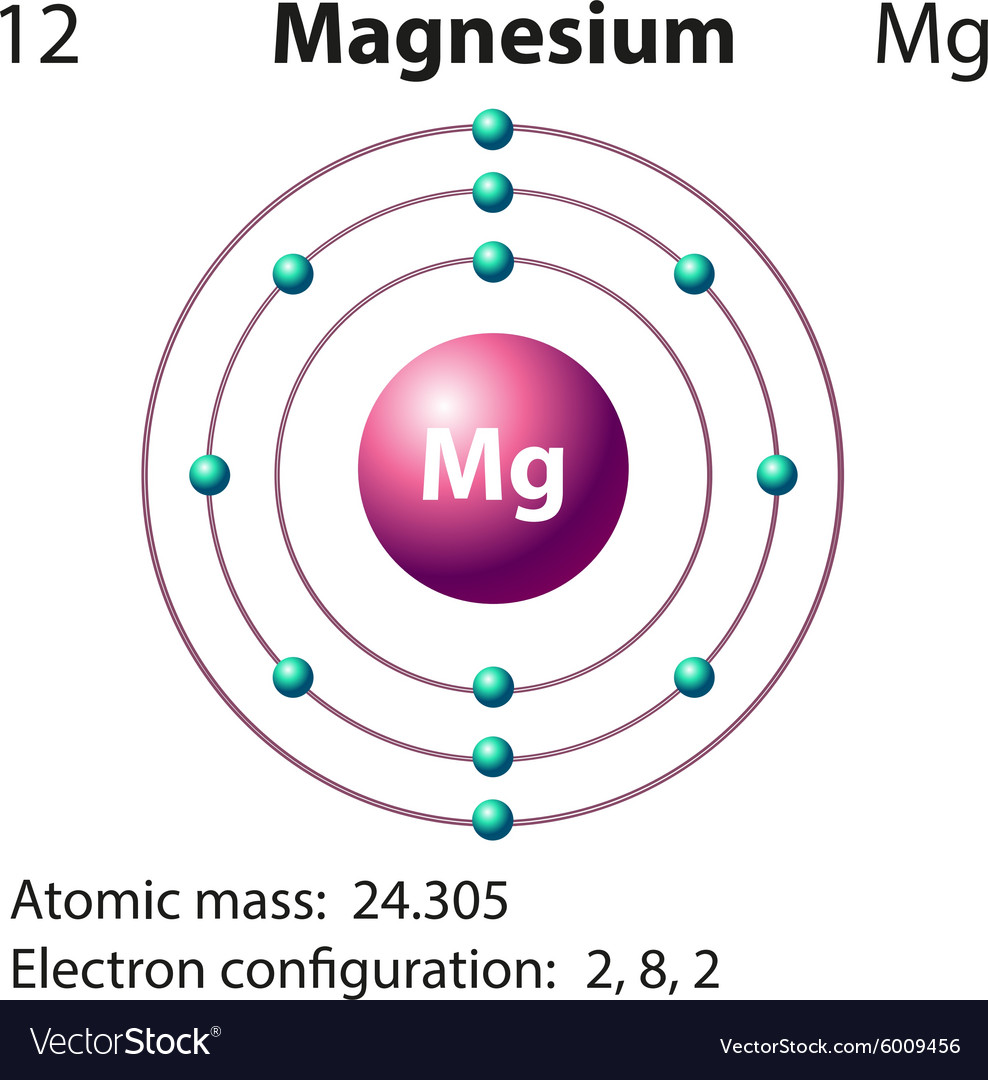
Pure polycrystalline magnesium is brittle and easily fractures along shear bands. Magnesium has the lowest melting (923 K (650 ☌)) and the lowest boiling point (1,363 K (1,090 ☌)) of all the alkaline earth metals. Įlemental magnesium is a gray-white lightweight metal, two-thirds the density of aluminium. Magnesium compounds are used medicinally as common laxatives and antacids (such as milk of magnesia), and to stabilize abnormal nerve excitation or blood vessel spasm in such conditions as eclampsia. Hundreds of enzymes require magnesium ions to function. Magnesium ions interact with polyphosphate compounds such as ATP, DNA, and RNA. This element is the eleventh most abundant element by mass in the human body and is essential to all cells and some 300 enzymes. It is the third most abundant element dissolved in seawater, after sodium and chlorine. Magnesium is the eighth most abundant element in the Earth's crust and the fourth most common element in the Earth (after iron, oxygen and silicon), making up 13% of the planet's mass and a large fraction of the planet's mantle. When such stars explode as supernovas, much of the magnesium is expelled into the interstellar medium where it may recycle into new star systems. In the cosmos, magnesium is produced in large, aging stars by the sequential addition of three helium nuclei to a carbon nucleus. It is less dense than aluminium and is used primarily as a component in strong and lightweight alloys that contain aluminium. The metal is obtained mainly by electrolysis of magnesium salts obtained from brine. The free metal burns with a brilliant-white light. It reacts readily with air to form a thin passivation coating of magnesium oxide that inhibits further corrosion of the metal. Like the other alkaline earth metals (group 2 of the periodic table) it occurs naturally only in combination with other elements and it almost always has an oxidation state of +2. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Magnesium is a chemical element with the symbol Mg and atomic number 12.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
